What Is A DMD? A Comprehensive Guide To Duchenne Muscular Dystrophy
What is a DMD? This simple, three-letter acronym opens the door to a complex and challenging medical reality for thousands of families worldwide. DMD stands for Duchenne Muscular Dystrophy, the most common and one of the most severe forms of muscular dystrophy. It is a progressive genetic disorder characterized by the gradual weakening and deterioration of skeletal muscles, which are the muscles used for movement. Understanding "what is a DMD" is the critical first step for anyone seeking to support a loved one, navigate a new diagnosis, or simply educate themselves about this condition. This guide will walk you through everything you need to know, from the genetic blueprint of the disease to the cutting-edge therapies offering new hope.
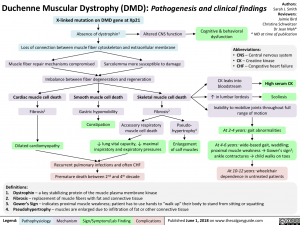
Duchenne Muscular Dystrophy is not just a muscle disease; it is a systemic condition that affects multiple body systems over time. It primarily impacts boys, with an incidence of about 1 in 3,500 to 5,000 live male births globally. The journey of DMD is marked by distinct stages, from early childhood motor delays to the loss of ambulation in adolescence and the involvement of cardiac and respiratory muscles in adulthood. While historically a life-limiting diagnosis, advancements in cardiac and respiratory care, coupled with revolutionary new treatments, are significantly changing the prognosis and quality of life for individuals with DMD. This article aims to demystify the condition, providing a clear, compassionate, and comprehensive overview.
What Exactly is Duchenne Muscular Dystrophy?
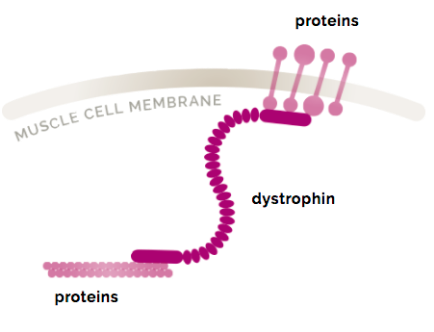
At its core, Duchenne Muscular Dystrophy is a genetic disorder caused by mutations in the DMD gene. This gene provides the instructions for making a crucial protein called dystrophin. Dystrophin acts as a shock absorber and structural anchor within muscle fibers, helping to keep muscle cell membranes intact during contraction and relaxation. When the DMD gene is mutated, dystrophin production is either severely reduced or absent entirely. Without this protective protein, muscle fibers become fragile, easily damaged, and progressively replaced by scar tissue and fat. This process of muscle degeneration and fibrosis is what leads to the characteristic muscle weakness.
The condition is named after Dr. Guillaume Duchenne, a French neurologist who first described the disease in detail in the 1860s. He was a pioneer in using electrical stimulation to study muscle function and meticulously documented the clinical progression in his patients. His work laid the foundation for understanding the disease that would eventually bear his name. DMD is classified as an X-linked recessive disorder. This means the gene mutation is located on the X chromosome. Since males have only one X chromosome, a single mutated copy of the gene is enough to cause the disease. Females, with two X chromosomes, are typically carriers; they usually have one functional copy of the gene and are rarely affected, though some can experience milder symptoms.
The Genetic Roots: How Does a Person Get DMD?
Understanding the inheritance pattern is key to answering "what is a DMD" from a familial perspective. Approximately two-thirds of DMD cases are inherited from a carrier mother. The remaining one-third result from a de novo (new) mutation, where the genetic change occurs spontaneously in the egg or sperm cell, or very early in fetal development. In these cases, the mother is not a carrier, and the risk of having another affected child is low, though there is a risk of germline mosaicism.
For families with a known history, genetic counseling is invaluable. A carrier mother has a 50% chance of passing the mutated gene to each son (who would have DMD) and a 50% chance of passing it to each daughter (who would become a carrier). Genetic testing can identify the specific mutation in the DMD gene in about 95% of cases. This precise diagnosis is essential not only for family planning but also for determining eligibility for emerging mutation-specific therapies, such as exon-skipping drugs, which are designed to work on specific genetic mutations. The complexity of genetic inheritance underscores why a simple blood test for the mother and the affected individual is the gold standard for confirmation.
Recognizing the Signs: The Symptom Journey of DMD
The symptoms of DMD typically appear in early childhood, usually between ages 2 and 5. The first signs are often developmental delays related to motor skills. Parents might notice:
- Difficulty running, jumping, or climbing stairs.
- Frequent falls and a tendency to toe-walk.
- Delayed ability to sit, stand, or walk independently.
- Larger-than-normal calf muscles (pseudohypertrophy), which are actually filled with fat and scar tissue, not strong muscle.
- Trouble getting up from the floor (using hands to "climb" up the legs—Gowers' sign).
As the child grows, muscle weakness progresses in a characteristic pattern, affecting the proximal muscles (those near the trunk) first—the hips, pelvis, thighs, and shoulders. By early adolescence, most boys with DMD lose the ability to walk and become fully wheelchair-dependent, typically between ages 10 and 12. The disease then progresses to the upper body, affecting arm and neck muscles. Critically, the heart muscle (cardiomyopathy) and breathing muscles are also affected, usually in the teenage years and beyond. Cardiac complications, particularly dilated cardiomyopathy, and respiratory insufficiency are now the leading causes of mortality in DMD. This progressive, multi-system nature of DMD necessitates a proactive, multidisciplinary care approach involving neurologists, cardiologists, pulmonologists, physical therapists, and orthopedic specialists.
How is DMD Diagnosed? From Suspicion to Confirmation
Diagnosis is a multi-step process that begins with clinical observation. A pediatrician or family doctor noticing the characteristic signs will typically order a creatine kinase (CK) test. CK is an enzyme released into the blood when muscle fibers are damaged. In DMD, CK levels are extremely elevated, often 10 to 100 times the normal range, even before significant weakness is apparent. This is a crucial early red flag.
If CK levels are high, the definitive diagnostic test is a muscle biopsy or, more commonly now, a genetic blood test. The muscle biopsy involves removing a small sample of muscle tissue to examine under a microscope for the absence of dystrophin protein and the presence of dystrophic changes. However, the genetic test is now the preferred first confirmatory test. It sequences the DMD gene to identify the specific mutation. This test is less invasive, provides a definitive diagnosis, and is necessary for accessing mutation-targeted therapies. Once diagnosed, a comprehensive baseline evaluation is conducted, including cardiac (echocardiogram, ECG) and pulmonary (pulmonary function tests) assessments to establish a starting point for ongoing monitoring and care.
Current Treatments and Management Strategies: A Multidisciplinary Battle
While there is currently no cure for DMD, treatment is a powerful combination of therapies aimed at slowing progression, managing symptoms, and maximizing quality of life and longevity. The cornerstone is corticosteroid therapy (prednisone or deflazacort). These drugs have been shown for decades to slow muscle degeneration, prolong the ability to walk, and delay the onset of scoliosis and cardiopulmonary complications. The timing, dosage, and management of steroid side effects (like weight gain, growth suppression, and bone density loss) are carefully balanced by a neuromuscular care team.
Beyond steroids, a robust supportive care regimen is non-negotiable:
- Physical and Occupational Therapy: Daily stretching to prevent contractures (permanent muscle shortening), low-impact exercises to maintain strength and cardiovascular health, and use of adaptive equipment.
- Cardiac Care: Regular monitoring with echocardiograms and ECGs. Medications like ACE inhibitors or beta-blockers are often started early to protect heart function.
- Respiratory Support: As breathing muscles weaken, non-invasive ventilation (BiPAP) is used, especially during sleep, to prevent respiratory failure.
- Orthopedic Interventions: Surgery for scoliosis (spinal fusion) and contracture release (Achilles tendon lengthening) can improve sitting balance, comfort, and pulmonary function.
- Nutritional Support: A balanced diet is vital, but calorie intake must be managed to prevent obesity, which exacerbates mobility and respiratory challenges.
The Hope on the Horizon: Mutation-Targeted and Gene Therapies
The landscape of DMD treatment is undergoing a revolution. For the first time, therapies are moving beyond symptom management to target the underlying genetic cause. Exon-skipping therapies (e.g., eteplirsen, golodirsen, viltolarsen) are FDA-approved antisense oligonucleotide drugs. They work by "skipping" over a faulty exon (a section of the DMD gene) during RNA processing, allowing the production of a shorter, but partially functional, dystrophin protein. This is akin to turning a "stop" sign into a "go" sign for protein production. These drugs are mutation-specific, meaning they only work for patients with specific, eligible mutations (about 10-15% of the DMD population).
Even more transformative are the new gene replacement therapies. In 2023, the FDA approved Elevidys (delandistrogene moxeparvovec), a one-time intravenous gene therapy for 4-5 year old ambulant boys with DMD. It uses a harmless virus (AAV) to deliver a miniaturized, functional copy of the DMD gene to muscle cells, enabling them to produce a shortened form of dystrophin. This represents a monumental shift from chronic treatment to a potential one-time intervention. Furthermore, gene editing technologies like CRISPR are in preclinical and early clinical stages, aiming to permanently correct the mutation within the DNA itself. These advances signal a new era where the question "what is a DMD" may soon be followed by "how can we fix it?"
Living with DMD: A Family's Journey of Resilience and Advocacy
A DMD diagnosis is a life-altering event for the entire family. It reshapes daily routines, financial planning, and emotional landscapes. Psychosocial support is a critical component of comprehensive care. Connecting with patient advocacy organizations like the Parent Project Muscular Dystrophy (PPMD), CureDuchenne, or Muscular Dystrophy Association (MDA) provides families with invaluable resources, community connections, and guidance on navigating healthcare systems and clinical trials.
Practical daily life involves meticulous planning. Home modifications (ramps, stairlifts, accessible bathrooms) become necessary. Educational plans (IEPs) must be developed to ensure the child's needs are met at school. As the individual with DMD transitions into adulthood, planning for higher education, vocational training, and independent living with appropriate support services becomes the focus. Caregiver burnout is a real and significant risk. It is essential for parents and caregivers to seek respite care, mental health support, and to build a network of trusted medical professionals and community resources. The journey is arduous, but it is also filled with moments of profound joy, resilience, and love. Many young men with DMD today are graduating high school, attending college, pursuing careers, and advocating for themselves and others, redefining what is possible.
Frequently Asked Questions About DMD
Q: Is Duchenne Muscular Dystrophy painful?
A: The disease process itself is not typically described as sharp or chronic pain. However, muscle cramps, joint stiffness from contractures, and discomfort from prolonged sitting can occur. These are managed with physical therapy, stretching, medications, and positioning.
Q: What is the life expectancy for someone with DMD?
A: Historically, life expectancy was in the late teens or early twenties. With modern, comprehensive cardiopulmonary care, many individuals are now living into their 30s, 40s, and beyond. Life expectancy continues to rise as treatments improve.
Q: Can girls get Duchenne Muscular Dystrophy?
A: It is extremely rare. Females with a single DMD gene mutation are usually asymptomatic carriers. However, due to a process called X-inactivation, some carrier females can experience mild muscle weakness, cramps, or cardiac issues later in life and should be monitored by a cardiologist.
Q: Are there any lifestyle or dietary restrictions?
A: There are no specific forbidden foods, but a balanced, nutrient-dense diet is crucial. Obesity must be avoided as extra weight severely strains weakened muscles and the respiratory system. Some families explore anti-inflammatory or antioxidant-rich diets, but any supplement regimen should be discussed with the care team.
Q: How can I support someone with DMD?
A: The best support is often practical and emotional. Offer specific help (e.g., driving to appointments, preparing a meal). Educate yourself on the disease. Listen without pity. Support family caregivers. Donate to reputable DMD research organizations. Most importantly, see the person, not just the disease.
Conclusion: Redefining the Narrative of DMD
So, what is a DMD? It is a complex genetic disorder defined by the absence of dystrophin, leading to progressive muscle degeneration. It is a diagnosis that brings immense challenges but also catalyzes extraordinary courage, scientific ingenuity, and community strength. It is a journey that begins with a genetic blueprint but is ultimately written by the individuals living with it, their families, and the global community of researchers and clinicians dedicated to finding treatments and a cure.
The story of DMD is being rewritten in real-time. From the foundational understanding of dystrophin's role to the approval of mutation-targeted exon-skippers and landmark gene therapies, the trajectory has shifted from one of inevitable decline to one of managed progression and tangible hope. While the road remains difficult, the future is brighter than it has ever been. By understanding the medical realities, embracing proactive multidisciplinary care, and supporting relentless research, we move closer to a world where Duchenne Muscular Dystrophy is no longer a devastating diagnosis, but a treatable, and eventually curable, condition. Knowledge is the first and most powerful weapon in that fight.