Integral Vs Peripheral Proteins: A Deep Dive Into Cellular Architecture And Function
Have you ever wondered how your cells know what to do? The answer lies in a bustling, microscopic metropolis inside every cell, where proteins are the citizens, each with a specific job. But how do these proteins get their assignments and know where to go? The distinction between integral vs peripheral proteins is fundamental to understanding cellular life. These two classes of membrane-associated proteins are like the architects and the mobile workforce of the cell's boundary, working in tandem to control what enters and exits, send signals, and maintain structure. Grasping their differences isn't just academic—it's crucial for understanding health, disease, and the development of life-saving medications.
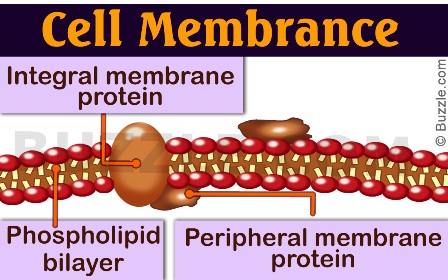
The cell membrane is not a simple barrier; it's a dynamic, fluid mosaic primarily composed of lipids and proteins. Proteins associated with this membrane are broadly categorized based on their relationship with the lipid bilayer. Integral proteins are permanently embedded within the membrane, often spanning its entire width, while peripheral proteins are temporarily attached to the membrane's surface, either to the lipid heads or to integral proteins. This seemingly simple classification has profound implications for their structure, function, and how we study them. In this comprehensive guide, we'll unravel the mysteries of integral and peripheral proteins, exploring their unique characteristics, vital roles, and why this distinction is a cornerstone of modern cell biology and biochemistry.
Defining the Players: What Are Integral and Peripheral Proteins?
Before we compare them, we must clearly define each player in this cellular drama. The classification hinges on the strength and nature of their association with the phospholipid bilayer.
Integral Proteins: The Embedded Gatekeepers
Integral membrane proteins (IMPs), also known as transmembrane proteins, are permanently attached to the membrane. Their defining feature is one or more hydrophobic domains that are embedded within the fatty acid core of the lipid bilayer. These hydrophobic regions are typically composed of 20-25 nonpolar amino acids, often arranged in alpha-helical segments or, less commonly, beta-barrel structures. Because their hydrophobic regions are sequestered from water, integral proteins cannot be separated from the membrane without disrupting the lipid bilayer itself, usually requiring harsh detergents or organic solvents to solubilize them.
They can be classified further based on their topology:
- Single-pass transmembrane proteins cross the membrane once.
- Multi-pass transmembrane proteins weave in and out multiple times (e.g., G-protein coupled receptors with 7 transmembrane helices).
- Integral monotopic proteins are embedded in only one leaflet of the bilayer but do not span it.
Peripheral Proteins: The Attached Assistants
Peripheral membrane proteins are temporarily associated with the membrane's surface. They are not embedded in the hydrophobic core. Instead, they bind reversibly to:
- The polar head groups of phospholipids via electrostatic interactions or hydrogen bonds.
- The hydrophilic domains of integral membrane proteins.
- Both, forming a bridge.
Because their attachment is non-covalent and relatively weak, peripheral proteins can be detached from the membrane using mild treatments like changing salt concentration, pH, or adding chelating agents (e.g., EDTA to remove bound ions). This reversibility is key to their function, allowing them to come and go as cellular signals dictate. They are often found on the cytoplasmic side of the plasma membrane but can also be on the extracellular face.
Structural Showdown: How Their Designs Dictate Their Duties
The fundamental structural differences between integral and peripheral proteins are a direct consequence of their relationship with the hydrophobic lipid core, and these differences dictate everything else.
The Hydrophobic Heart of Integral Proteins
The hydrophobic effect is the driving force here. The interior of the lipid bilayer is an energetically unfavorable environment for polar or charged molecules. Integral proteins have evolved hydrophobic transmembrane domains that are energetically compatible with this environment. These domains are the "anchor." Flanking these hydrophobic regions are hydrophilic domains that extend into the aqueous environments on either side of the membrane—the extracellular space and the cytoplasm. This creates a protein with a distinct orientation, or topology, where the N- and C-termini are on specific sides. This asymmetric insertion is critical for function; for example, a receptor's ligand-binding site must face outward.
The amino acid composition of transmembrane helices is telling. They are enriched in hydrophobic residues like leucine, isoleucine, valine, phenylalanine, and methionine, and depleted in polar residues like lysine, arginine, glutamate, and aspartate. However, occasional polar residues within the helix are not mistakes; they often form crucial functional sites, such as the pore-lining residues in ion channels or the proton shuttle in bacteriorhodopsin.
The Dynamic Attachment of Peripheral Proteins
Peripheral proteins lack these long hydrophobic stretches. Their sequences are rich in polar and charged amino acids, making them soluble in the cytosol. Their membrane attachment is mediated by specific binding domains. For example:
- PH domains bind to phosphoinositides (a type of phospholipid).
- C2 domains bind to phospholipids in a calcium-dependent manner.
- Annexins bind to phospholipids and calcium.
- Many bind directly to specific cytoplasmic domains of integral proteins, like the interaction between spectrin (peripheral) and ankyrin (adaptor protein linked to integral channels).
This dynamic, reversible binding is perfect for regulatory roles. A peripheral signaling protein can be recruited to the membrane only when a specific lipid second messenger is produced or when a calcium influx occurs, providing precise spatial and temporal control.
Functional Divide: What Each Protein Type Does Best
Their structural blueprints assign them to different, yet often complementary, functional zones within the cell.
Integral Proteins in Action: Transport, Signaling, and Adhesion
Integral proteins are the workhorses of the membrane's active functions.
- Channels and Pores: Form hydrophilic tunnels for the passive diffusion of specific ions (e.g., potassium channels, sodium channels) or molecules (e.g., aquaporins for water). Their gates can open and close in response to voltage, ligands, or mechanical stress.
- Transporters/Carriers: Undergo conformational changes to move substances against their concentration gradient (active transport) or facilitate facilitated diffusion. The sodium-potassium pump (Na+/K+-ATPase) is a classic example, using ATP to maintain the electrochemical gradient essential for nerve impulses.
- Receptors: Have extracellular domains that bind specific ligands (hormones, neurotransmitters, growth factors). This binding triggers a conformational change that initiates a signaling cascade inside the cell. G-protein coupled receptors (GPCRs) and receptor tyrosine kinases (RTKs) are major drug targets.
- Enzymes: Catalyze reactions at the membrane interface, like gamma-secretase involved in Alzheimer's disease pathology.
- Cell Adhesion Molecules (CAMs): Like integrins and cadherins, they mediate cell-cell and cell-extracellular matrix adhesion, critical for tissue formation and immune responses.
Peripheral Proteins: The Cellular Support Crew
Peripheral proteins are the versatile managers, anchors, and signal modulators.
- Cytoskeletal Anchors: Link the membrane to the internal cytoskeleton, providing mechanical strength and shape. Spectrin (linked by ankyrin to integral Na+ channels) forms a mesh under the red blood cell membrane. ERM proteins (ezrin, radixin, moesin) connect the plasma membrane to actin filaments.
- Signaling Components: Many are enzymes themselves (kinases, phosphatases) or regulators of signaling pathways. For instance, Src family kinases are peripheral proteins that phosphorylate integral receptors and other substrates upon recruitment to the membrane.
- Shape and Movement: Proteins like dynamin (a GTPase) assemble around the neck of budding vesicles to pinch them off from the membrane (endocytosis).
- Enzyme Regulation: Some peripheral proteins are regulatory subunits that bind to integral enzymes to activate or inhibit them.
Real-World Examples: From Ion Channels to Cytoskeletal Links
Let's make this concrete with specific, well-studied examples.
Integral Protein Examples:
- Potassium Channel (Kv1.2): A multi-pass alpha-helical channel that allows K+ ions to exit the cell down their electrochemical gradient, crucial for setting the resting membrane potential.
- CFTR (Cystic Fibrosis Transmembrane Conductance Regulator): An ABC transporter that functions as a chloride channel. Mutations in this integral protein cause cystic fibrosis.
- Beta-2 Adrenergic Receptor: A GPCR that binds adrenaline, triggering heart rate increase and other fight-or-flight responses. It's a prime target for asthma and cardiovascular drugs.
- Band 3 Protein (Anion Exchanger 1): A major integral protein in red blood cells that exchanges Cl- for HCO3- across the membrane, vital for CO2 transport.
Peripheral Protein Examples:
- Spectrin: Forms a 2D lattice beneath the plasma membrane in erythrocytes, providing flexibility and durability. Defects cause hereditary spherocytosis.
- G Proteins (Heterotrimeric): Bind to activated GPCRs (integral). The Gα subunit exchanges GDP for GTP and dissociates to regulate effectors like adenylyl cyclase.
- Phospholipase C (PLC): A peripheral enzyme that, upon activation by RTKs or GPCRs, hydrolyzes PIP2 into IP3 and DAG, two key second messengers.
- Ankyrin: An adaptor protein that binds the cytoplasmic domain of the integral Na+ channel (in neurons) and links it to the spectrin cytoskeleton.
How Scientists Study These Proteins: Techniques and Challenges
Studying these proteins presents unique challenges due to their hydrophobic nature and membrane environment.
For Integral Proteins:
- X-ray Crystallography & Cryo-EM: Require isolating and purifying the protein in detergent micelles or lipid nanodiscs to mimic the membrane environment. Cryo-electron microscopy (cryo-EM) has revolutionized this field, allowing high-resolution structures of large complexes in near-native lipid environments.
- Fluorescence Recovery After Photobleaching (FRAP): Measures the mobility of integral proteins within the fluid membrane.
- Site-Directed Mutagenesis: Changing specific amino acids in transmembrane domains to probe structure-function relationships.
For Peripheral Proteins:
- Surface Plasmon Resonance (SPR) & Isothermal Titration Calorimetry (ITC): Quantify the binding affinity and kinetics between a peripheral protein and its lipid or integral protein partner.
- Sedimentation Assays: Separate membrane-bound from soluble protein by centrifugation after different extraction treatments.
- Live-Cell Imaging: Tagging with fluorescent proteins (e.g., GFP) to visualize recruitment to and dissociation from the plasma membrane in real-time in response to stimuli.
The major challenge for both is reconstituting them into a functional, native-like lipid environment for in vitro study, as their behavior can be drastically different in detergent versus a real bilayer.
Clinical Connections: When Protein Function Goes Awry
Dysfunction in membrane proteins is at the root of countless diseases, making them the targets of ~50% of all modern pharmaceuticals.
Integral Protein Disorders:
- Cystic Fibrosis: Caused by mutations in the CFTR chloride channel (integral), leading to thick mucus in lungs and pancreas.
- Long QT Syndrome: Mutations in cardiac potassium or sodium channels (integral) disrupt heart rhythm, potentially causing sudden death.
- Familial Hypercholesterolemia: Defects in the LDL receptor (an integral receptor) impair cholesterol uptake.
- Many Cancers: Often involve overexpressed or mutated receptor tyrosine kinases (e.g., HER2/neu) or GPCRs that drive uncontrolled proliferation.
Peripheral Protein Disorders:
- Hereditary Spherocytosis: Mutations in spectrin, ankyrin, or band 3 (which interacts with both integral and peripheral) cause fragile, spherical red blood cells.
- Neurological Disorders: Mutations in peripheral scaffolding proteins like dystrophin (linked to the integral dystroglycan complex) cause Duchenne muscular dystrophy. Defects in peripheral signaling adaptors like GRB2 are implicated in developmental disorders.
- Immune Deficiencies: Mutations in peripheral signaling molecules like ZAP-70 (a kinase recruited to the T-cell receptor complex) cause severe combined immunodeficiency.
Understanding the integral vs peripheral distinction is therefore not just about basic science; it's the language of pathophysiology and therapeutics.
Evolutionary Perspective: Why Two Types?
The existence of both protein types is a elegant evolutionary solution to the challenge of building a selective barrier.
- Integral proteins likely evolved from soluble proteins that acquired long hydrophobic helices, allowing them to embed and create permanent conduits or sensors within the barrier itself. Their structure is intrinsically stable within the hydrophobic core.
- Peripheral proteins represent a more flexible, modular system. They can be rapidly recruited from the cytosol as needed, allowing for dynamic, signal-dependent control without the need to synthesize and insert a new transmembrane protein. This system allows for complex, tunable signaling networks. The two systems co-evolved, with many peripheral proteins developing specific interfaces to interact with particular integral proteins, creating intricate, regulated machines like the eukaryotic ribosome (attached to the ER membrane via the Signal Recognition Particle and its receptor, both peripheral and integral components).
Conclusion: A Symphony of Cellular Architecture
The dichotomy of integral vs peripheral proteins reveals a fundamental principle of cellular design: permanence versus flexibility. Integral proteins are the permanent, structural, and functional pillars of the membrane, forming the channels, pumps, and receptors that define the cell's interaction with its world. They are the architecture. Peripheral proteins are the dynamic, responsive workforce—the anchors, regulators, and signal transducers that adapt the cell's behavior in real-time. They are the management and maintenance crew.
One cannot function optimally without the other. Integral proteins provide the docking stations and platforms; peripheral proteins provide the regulatory switches and mechanical links. Together, they create a responsive, intelligent boundary that is far more than a passive sack. This intricate partnership is why mutations in either class can have such devastating effects, and why they remain such a fertile ground for drug discovery. The next time you consider a cell, picture this bustling interface: a sea of lipids with anchored integral sentries and a cloud of peripheral associates coming and going, all in a meticulously choreographed dance that is the very essence of life. Understanding this dance is the key to understanding biology itself.