Sodium Protons Neutrons Electrons: The Atomic Blueprint Of A Chemical Giant
Have you ever wondered what makes a pinch of table salt or the bright glow of a street light possible? The answer lies hidden within the heart of a single atom, governed by the precise dance of sodium protons neutrons electrons. These three subatomic particles are the fundamental building blocks that define sodium's identity, its explosive reactivity, and its gentle, life-sustaining role in our bodies. Understanding their numbers and arrangement isn't just academic; it's the key to unlocking the story of one of Earth's most essential and paradoxical elements. This comprehensive guide will deconstruct sodium atom by particle, revealing how the count of its protons, neutrons, and electrons orchestrates everything from the periodic table to the chemistry of life itself.
The Foundation: What Are Protons, Neutrons, and Electrons?
Before we zero in on sodium, let's establish the universal rules of atomic architecture. Every atom is a tiny solar system, but instead of planets, it has a dense nucleus orbited by electrons. The protons and neutrons reside in the nucleus, while the electrons whiz around in cloud-like shells.
- Protons carry a positive electrical charge. The number of protons in an atom's nucleus is its atomic number (Z), and this number is absolute and unchanging for a given element. It is the element's identity card. Change the proton count, and you have a different element entirely.
- Neutrons are electrically neutral, as their name suggests. They add mass to the atom and, crucially, help hold the positively charged protons together in the nucleus by providing the strong nuclear force that overcomes electrostatic repulsion. The number of neutrons can vary, leading to isotopes of the same element.
- Electrons are negatively charged particles with negligible mass. They exist in specific energy levels or shells around the nucleus. In a neutral atom, the number of electrons equals the number of protons. It's the arrangement and behavior of these outermost electrons—the valence electrons—that dictate an element's chemical personality and reactivity.
This triad—protons, neutrons, electrons—forms the complete blueprint. For sodium, this blueprint is particularly fascinating due to its single, eager valence electron.
Sodium's Atomic Signature: The Proton Count
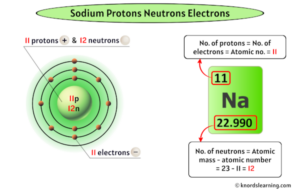
The story of sodium's atomic identity begins and ends with its proton count. Sodium has 11 protons. This is not an estimate or an average; it is the definitive, non-negotiable fact that places sodium squarely at atomic number 11 on the periodic table.
Why Sodium's Atomic Number Matters
This number 11 is sodium's DNA. It determines:
- Elemental Identity: Any atom with 11 protons is sodium (Na, from Natrium). An atom with 10 protons is neon, and with 12 protons is magnesium. The periodic table is essentially a list of proton counts.
- Electron Count in a Neutral Atom: In a neutral, stable sodium atom, the number of orbiting electrons must equal the number of protons to balance the charge. Therefore, a neutral sodium atom has 11 electrons.
- Chemical Behavior: The proton count dictates the number of core electrons and, most importantly, the number of valence electrons. For sodium, with 11 total electrons, the electron configuration is 2-8-1. That single electron in the third shell makes sodium highly reactive, as it "wants" to lose that one electron to achieve a stable, full outer shell like neon.
This is why sodium is a classic alkali metal. Its entire reactive personality stems from having just one electron to shed, a direct consequence of having 11 protons organizing 11 electrons into that 2-8-1 pattern.
The Mass Factor: Calculating Sodium's Neutrons
While protons define the element, neutrons define the specific version or isotope of that element. The mass number (A) of an atom is the total count of protons and neutrons in its nucleus. For the most common and stable form of sodium, we look at its standard atomic weight.
The Most Abundant Isotope: Sodium-23
The atomic mass of sodium listed on the periodic table is approximately 22.99 atomic mass units (amu). This isn't a whole number because it's a weighted average of all naturally occurring isotopes. However, the most abundant and only stable isotope of sodium is sodium-23.
Here’s the simple calculation for sodium-23:
- Mass Number (A) = 23
- Atomic Number (Z) = 11 (protons)
- Number of Neutrons = A - Z = 23 - 11 = 12 neutrons
So, the most common sodium atom you encounter has a nucleus packed with 11 protons and 12 neutrons, giving it a mass number of 23. This isotope makes up 100% of naturally occurring sodium.
The Concept of Isotopes: Sodium-22 and Sodium-24
While sodium-23 is stable, other isotopes exist, created in nuclear reactions. For example:
- Sodium-22: Has 11 protons and 11 neutrons (22 - 11 = 11). It is radioactive, decaying by positron emission, and is used in medical PET scans.
- Sodium-24: Has 11 protons and 13 neutrons (24 - 11 = 13). It is also radioactive and a crucial byproduct in nuclear reactors, used to trace coolant leaks.
The existence of these isotopes proves that while the proton count is sacred, the neutron count can vary, affecting the atom's mass and stability but not its fundamental chemical identity as sodium.
Electron Configuration: The Key to Sodium's Reactivity
With 11 protons defining the nucleus, we now distribute 11 electrons around it. This distribution follows strict quantum mechanical rules and is written as sodium's electron configuration: 1s² 2s² 2p⁶ 3s¹.
Let's break down what this sequence of numbers and letters means for sodium's behavior:
Shell-by-Shell Breakdown
- First Shell (n=1): Holds up to 2 electrons. Sodium fills this completely: 1s².
- Second Shell (n=2): Holds up to 8 electrons. Sodium fills this completely: 2s² 2p⁶. This gives sodium the stable electron configuration of neon, a noble gas, for its inner core.
- Third Shell (n=3): This is where the action is. Sodium places its 11th electron here in the 3s orbital: 3s¹.
That lone 3s¹ electron is sodium's chemical destiny. It is far from the nucleus, shielded by the full inner shells, and only loosely held by the positive charge of the 11 protons. This makes it incredibly easy for sodium to lose this one electron, transforming into a Na⁺ cation with a stable neon-like electron configuration (2-8).
The Noble Gas Connection and the Octet Rule
This drive to achieve a full outer shell of 8 electrons (the octet rule) is the engine of sodium's chemistry. By losing its one valence electron, sodium achieves a stable, low-energy state. This is why sodium is never found free in nature; it's always bonded as an ion, most famously paired with chloride (Cl⁻) in sodium chloride (NaCl), common table salt. The electrostatic attraction between Na⁺ and Cl⁻ forms the classic ionic bond.
Position in the Periodic Table: A Family Portrait
Sodium's location on the periodic table is a direct map of its proton, neutron, and electron story.
Group 1: The Alkali Metals
Sodium sits in Group 1 (IA), the first column. Every element in this group has a single electron in its outermost s-orbital (ns¹ configuration). This means:
- Lithium (Li): 3 protons, configuration 1s² 2s¹
- Sodium (Na): 11 protons, configuration [Ne] 3s¹
- Potassium (K): 19 protons, configuration [Ar] 4s¹
- ...and so on down to Francium.
They share extreme reactivity, softness, low density, and the characteristic +1 oxidation state. Their single valence electron is given up with thrilling ease.
Period 3: The Third Row
Sodium is in Period 3. This means its outermost electrons are in the third principal energy level (n=3). Moving across Period 3 from sodium to argon, electrons are added to the same third shell, filling the 3s and 3p orbitals. Sodium, with its 3s¹, is the first element in this row, starting the pattern of filling the third shell.
The s-Block
Group 1 and 2 elements (alkali and alkaline earth metals) form the s-block of the periodic table, named for the s-orbital that houses their valence electrons. Sodium's entire chemical character is defined by that one electron in a 3s orbital.
Chemical Properties in Action: From Salt to Street Lights
The atomic structure we've dissected translates directly into the tangible, often dramatic, properties of elemental sodium and its compounds.
Physical Properties of Elemental Sodium
- Appearance: A soft, silvery-white metal that can be cut with a knife. It's so soft you can mold it.
- Reactivity with Air/Water: This is the most dramatic demonstration. Exposed to air, it quickly tarnishes as it reacts with oxygen and moisture. Dropped in water, it skitters violently, fizzing, hissing, and often bursting into flame (or even exploding) as it reacts exothermically:
2Na + 2H₂O → 2NaOH + H₂(g). The hydrogen gas produced can ignite from the heat of the reaction. This is why sodium is stored under mineral oil. - Density: Less dense than water (0.97 g/cm³), which is why it floats and reacts on the water's surface.
The Gentle Giant: Sodium Ions in Biology
The violent reactivity of pure sodium contrasts sharply with the peaceful role of the sodium ion (Na⁺).
- Nerve Impulse Transmission: The flow of Na⁺ ions across nerve cell membranes generates electrical signals. This is fundamental to all thought, movement, and sensation.
- Fluid Balance: Na⁺ is the primary cation in extracellular fluid, regulating blood pressure, blood volume, and pH balance.
- Muscle Contraction: Sodium influx triggers the electrical changes that lead to muscle fiber contraction.
Our bodies have intricate pumps (Na⁺/K⁺-ATPase) that use energy to maintain a high concentration of Na⁺ outside cells and K⁺ inside, creating a critical electrochemical gradient.
Industrial and Everyday Applications
- Table Salt (NaCl): The most familiar sodium compound. Used for seasoning, food preservation, and as a source of sodium ions.
- Sodium Vapor Lamps: The iconic orange-yellow street lights. Electricity passed through a sodium vapor excites the electrons; when they fall back to lower energy levels, they emit that characteristic monochromatic light, incredibly efficient for highways.
- Metallurgy: Sodium is used as a reducing agent to extract metals like titanium and zirconium from their chlorides.
- Chemical Feedstock: Sodium hydroxide (NaOH, lye/caustic soda) and sodium carbonate (Na₂CO₃, soda ash) are billion-ton-per-year industrial chemicals used in paper, soap, glass, and water treatment.
- Nuclear Reactors: Liquid sodium is used as a coolant in some fast-breeder nuclear reactors due to its excellent heat transfer properties and low neutron absorption.
Safety First: Handling the Reactive Metal
The dramatic reactivity of elemental sodium demands respect.
- Storage: Always under an inert liquid like mineral oil or kerosene to exclude air and moisture.
- Fire Response:Never use water! This intensifies the fire and can cause explosions. Use a Class D fire extinguisher specifically for metal fires (e.g., dry powder like Met-L-X or graphite-based).
- Biological Toxicity: Pure sodium metal is corrosive and causes severe chemical burns. However, sodium ions (Na⁺) are essential nutrients. The danger with salt (NaCl) is overconsumption. The WHO recommends less than 2,000 mg of sodium per day (about 5g of salt). High intake is linked to hypertension, heart disease, and stroke. The average person in many countries consumes over 3,400 mg daily.
Debunking Myths: Common Questions About Sodium's Atoms
Q1: Does sodium always have 11 electrons?
- A: Not always. In its neutral, elemental form, yes. But sodium almost instantly loses its valence electron to form the Na⁺ ion, which has only 10 electrons. This ion is what's found in salt and our bodies.
Q2: Why is the atomic mass 22.99 and not a whole number?
- A: Because it's an average. While sodium-23 (11p, 12n) is 100% abundant and stable, the atomic mass listed is the weighted average of all isotopes' masses (considering their natural abundance). The slight fractional mass comes from the mass defect (E=mc²) where nuclear binding energy converts a tiny amount of mass.
Q3: Can you have an atom with 11 protons and 10 neutrons?
- A: Yes, that's the radioactive isotope sodium-21 (11p, 10n). It's highly unstable with a half-life of about 22.5 seconds, decaying via proton emission. It doesn't occur naturally.
Q4: Is the number of neutrons always 12?
- A: For stable, naturally occurring sodium, yes. Sodium-23 is the only stable isotope. All other isotopes (like Na-22, Na-24) are radioactive and have either 11 or 13+ neutrons.
Conclusion: The Power of the Particle Count
The seemingly simple question "how many protons, neutrons, and electrons does sodium have?" opens a door to the profound logic of the atomic world. Sodium's 11 protons cement its identity as a reactive alkali metal. Its 12 neutrons (in the stable form) provide the nuclear mass without altering its chemical soul. Its 11 electrons, configured as 2-8-1, with a single, vulnerable electron in the outer shell, dictate its explosive reactivity and its ultimate transformation into the stable, life-giving Na⁺ ion.
From the violent fizz in a water droplet to the silent pulse of a nerve cell, from the glow of a city street to the seasoning on your fries, the story of sodium is written in the numbers of its subatomic particles. Understanding this atomic blueprint—sodium protons neutrons electrons—isn't just about memorizing figures. It's about comprehending the fundamental code that turns simple particles into the diverse and vital chemistry that shapes our world. The next time you see a street lamp or shake salt onto your food, you'll know the true architects of that moment: a nucleus of 11 protons and 12 neutrons, and an electron cloud arranged in a perfect, reactive 2-8-1 pattern.