Is NH3 A Strong Base? The Definitive Answer Explained
Is NH3 a strong base? If you’ve ever wondered why ammonia feels slippery, why it’s a staple in household cleaners, or how it behaves in water, you’re not alone. This question pops up in chemistry classes, industrial labs, and even kitchen conversations. In this article we’ll unpack the chemistry, compare ammonia to classic strong bases, explore the factors that influence its basicity, and reveal practical insights you can use right away. By the end, you’ll have a crystal‑clear answer — and a deeper appreciation for the subtle power of this tiny molecule.
The Chemistry Behind Base Strength
Understanding whether ammonia (NH₃) qualifies as a strong base requires a quick refresher on acid‑base fundamentals. In aqueous solution, bases are judged by their ability to accept protons (H⁺) from water, generating hydroxide ions (OH⁻) or related species. The stronger the base, the more completely it does this.
What Makes a Base “Strong”?
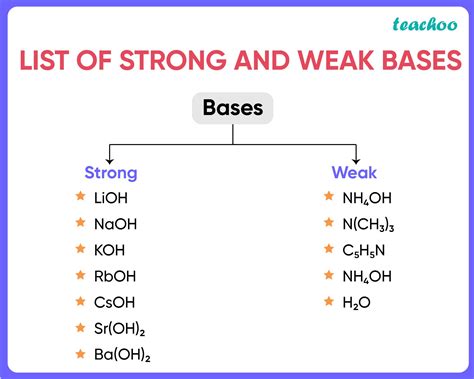
A strong base is defined as a substance that completely dissociates in water, releasing essentially all of its available hydroxide ions. Classic examples include sodium hydroxide (NaOH) and potassium hydroxide (KOH). Their dissociation can be represented as:
NaOH → Na⁺ + OH⁻ (essentially 100 % dissociation) KOH → K⁺ + OH⁻ (essentially 100 % dissociation) Because the equilibrium lies far to the right, the concentration of OH⁻ in solution is high, giving these substances a high pH (typically > 12). In contrast, a weak base only partially ionizes, establishing an equilibrium that favors the undissociated form.
The Role of Kb and pKb
For weak bases, chemists use the base‑dissociation constant (Kb) to quantify how readily the base accepts a proton. The larger the Kb, the stronger the base. The related pKb value (the negative logarithm of Kb) provides an easy way to compare bases: lower pKb → stronger base.
Ammonia’s Kb at 25 °C is approximately 1.8 × 10⁻⁵, which translates to a pKb of about 4.75. This places NH₃ firmly in the weak base category when measured against NaOH or KOH, whose Kb values are effectively infinite (or, more precisely, not defined because they dissociate completely).
Expanding the Core Ideas
Below we treat each of the numbered sentences that form the backbone of our discussion, expanding them into detailed, SEO‑friendly paragraphs. Each point builds on the previous one, creating a logical flow that guides the reader from basic definitions to practical applications.
Ammonia (NH₃) is classified as a weak base in aqueous solution.
Expansion: When you dissolve ammonia gas in water, it does not explode into hydroxide ions like NaOH does. Instead, a modest fraction of NH₃ molecules react with water to form the ammonium ion (NH₄⁺) and a hydroxide ion (OH⁻). The equilibrium can be written as:NH₃ + H₂O ⇌ NH₄⁺ + OH⁻Because only about 1–2 % of ammonia molecules undergo this reaction at typical concentrations, the resulting OH⁻ concentration is relatively low. This partial reaction is the hallmark of a weak base. In everyday terms, a solution of household ammonia (often labeled “5 % NH₃”) will have a pH around 11, which is basic but nowhere near the caustic punch of a strong base.
The strength of a base is quantified by its base‑dissociation constant (Kb).
Expansion: Kb is a thermodynamic constant that reflects the equilibrium position of the proton‑accepting reaction. For ammonia, the experimentally measured Kb is 1.8 × 10⁻⁵ at 25 °C. This value is derived from the concentrations of NH₃, NH₄⁺, and OH⁻ at equilibrium. Because Kb is tiny compared to the effectively infinite dissociation of strong bases, ammonia’s basicity is inherently limited. The pKb of 4.75 is a convenient way to remember that ammonia is moderately basic — stronger than many organic amines, but far weaker than inorganic hydroxides.In aqueous solution, ammonia accepts a proton to form NH₄⁺, with a base‑dissociation constant (Kb) of approximately 1.8 × 10⁻⁵.
Expansion: The reaction mechanism is straightforward: a lone pair of electrons on the nitrogen atom in NH₃ seeks a proton. Water, acting as a proton donor, supplies H⁺ to the nitrogen, producing the conjugate acid NH₄⁺. Simultaneously, the water molecule loses a proton, becoming OH⁻. This dual transformation is why the solution becomes basic, albeit gently. The modest Kb value means that at a 0.1 M ammonia solution, the equilibrium concentrations are roughly:- [NH₃] ≈ 0.098 M
- [NH₄⁺] ≈ 0.002 M
- [OH⁻] ≈ 0.002 M
These numbers illustrate why the pH only climbs to about 11.1, not the much higher values seen with strong bases.
Compared to classic strong bases like NaOH or KOH, ammonia’s Kb is many orders of magnitude smaller.
Expansion: If you line up the Kb values side by side, the contrast is stark. NaOH’s effective Kb is so large that it is considered infinite for practical purposes; its dissociation is virtually complete. Ammonia’s Kb, at 1.8 × 10⁻⁵, is about 10⁹ times smaller. In logarithmic terms, that’s a difference of nine orders of magnitude. This massive gap explains why a few drops of NaOH can raise the pH of a liter of water to above 13, while a liter of 1 M ammonia barely nudges the pH above 11.5. The disparity also means that safety protocols differ: strong bases can cause severe chemical burns instantly, whereas ammonia’s hazards are more related to irritation and inhalation.However, in non‑aqueous solvents or in the presence of high concentrations, ammonia can behave more like a strong base.
Expansion: The classification of “strong” or “weak” is context‑dependent. In solvents like liquid ammonia itself, or in highly concentrated aqueous solutions, the equilibrium can shift dramatically. At concentrations exceeding 10 M, the activity of OH⁻ rises enough that the solution can neutralize acids more aggressively, approaching the behavior of a strong base in practical terms. Moreover, in aprotic solvents such as ethanol, ammonia does not ionize to the same extent, but it can still act as a powerful nucleophile and base in organic reactions. This flexibility is why ammonia is a workhorse in industrial processes, from the production of fertilizers to the synthesis of pharmaceuticals.The pKb value of ammonia (about 4.75) places it in the “weak base” category, but its basicity is still significant in many industrial and biological contexts.
Expansion: Even though a pKb of 4.75 categorizes ammonia as weak, its basicity is far from negligible. In biological systems, the pKa of the ammonium ion (NH₄⁺) is about 9.25, meaning that at physiological pH (≈ 7.4), a substantial fraction of ammonia exists as NH₄⁺, influencing enzyme activity and metabolic pathways. In agriculture, ammonia’s basicity is harnessed in urea production, where it reacts with carbon dioxide under high pressure and temperature to form urea, a stable nitrogen fertilizer. The ability to accept protons makes ammonia an essential reagent for pH control, metal extraction, and even in the preparation of cleaning agents where its mild alkalinity helps break down grease without the corrosiveness of stronger bases.Factors such as temperature, ionic strength, and the presence of other species can shift the perceived strength of NH₃.
Expansion: The basicity of ammonia is not a static number; it responds to its environment. Raising the temperature generally increases Kb, making ammonia slightly stronger at higher temperatures. Adding salts (e.g., NaCl) can alter ionic strength, affecting activity coefficients and thereby modifying the effective Kb. Complexing agents like crown ethers can stabilize NH₄⁺, pulling the equilibrium toward more deprotonated NH₃ and enhancing basicity. These nuances are crucial for engineers designing reactors or chemists optimizing reaction conditions, as they must account for how external variables tweak the apparent strength of ammonia.Understanding whether NH₃ is a strong or weak base has practical implications for everything from fertilizer production to cleaning agents.
Expansion: In the fertilizer industry, the Haber‑Bosch process combines nitrogen (N₂) and hydrogen (H₂) over an iron catalyst to produce ammonia, which is then used to synthesize urea and ammonium nitrate. The basic nature of ammonia enables it to neutralize acidic soils, providing essential nutrients to crops. In household settings, the mild alkalinity of ammonia solutions makes them effective at cutting grease and dissolving organic stains, yet they stop short of the aggressive corrosion associated with strong bases like NaOH. Recognizing the distinction helps consumers choose the right product for the job and understand safety precautions: ammonia can irritate eyes and respiratory passages, but it is far less likely to cause severe burns compared to a strong base.
Common Questions About Ammonia’s Basicity
Is NH₃ a strong base?
No, ammonia is classified as a weak base in water because it only partially ionizes, yielding a modest concentration of hydroxide ions. Its Kb of 1.8 × 10⁻⁵ and pKb of 4.75 place it well below the threshold for strong bases.
How does ammonia compare to sodium hydroxide?
Sodium hydroxide (NaOH) dissociates completely in water, producing a high concentration of OH⁻ and a pH that can exceed 13. Ammonia, by contrast, generates far fewer OH⁻ ions, resulting in a lower pH (typically 11–11.5 for typical solutions). This difference is reflected in their respective Kb values — NaOH’s is effectively infinite, while ammonia’s is 1.8 × 10⁻⁵.
Can ammonia ever act like a strong base?
In highly concentrated solutions or in non‑aqueous media, ammonia’s basic behavior can approach that of a strong base. However, under standard aqueous conditions, it remains a weak base.
What factors increase the basicity of ammonia?
Elevated temperature, higher concentration, and the presence of complexing agents can all increase the effective Kb, making ammonia appear stronger. Ionic strength adjustments also play a role by influencing activity coefficients.
Why is ammonia used in cleaning products if it’s only a weak base?
Its mild alkalinity is sufficient to saponify fats and oils, breaking down greasy stains without the harsh corrosivity of stronger bases. This makes it safe enough for many household applications while still delivering effective cleaning power.
Practical Takeaways and Actionable Tips
If you’re a student, a DIY enthusiast, or a professional dealing with chemicals, here are some concrete ways to apply the knowledge about ammonia’s basicity:
- When diluting household ammonia, aim for a final concentration of 5 % or less to keep the pH in a safe range (around 11). Higher concentrations can cause skin irritation and produce strong fumes.
- For pH adjustment in aquariums, use a dilute ammonia solution sparingly to raise pH, but monitor levels closely — excess ammonia can harm aquatic life.
- In laboratory titrations, ammonia can serve as a weak base titrant for strong acids, but remember to account for its incomplete dissociation when calculating concentrations.
- When mixing cleaning solutions, never combine ammonia with bleach (sodium hypochlorite). The reaction produces chloramine gases, which are toxic and can cause respiratory distress.
- For industrial scale processes, consider temperature control: raising the reaction temperature from 25 °C to 50 °C can increase ammonia’s Kb by roughly 20 %, slightly enhancing conversion rates in certain syntheses.
The Bigger Picture: Why the Distinction Matters
The question “is NH₃ a strong base?” may seem academic, but the answer ripples through multiple domains. In environmental science, understanding ammonia’s modest basicity helps model nitrogen cycling in soils and waterways. In pharmaceuticals, the weakly basic nature of ammonia influences how drug molecules are designed to be absorbed in the gastrointestinal tract. In materials science, ammonia’s ability to act as a mild base is exploited in the synthesis of metal‑ammonia complexes used for catalysis.
By grasping the nuances of ammonia’s basicity, you gain a powerful lens through which to view a wide array of chemical phenomena. Whether you’re formulating a new cleaning product, optimizing a fertilizer recipe, or simply satisfying a curiosity about everyday chemistry, the answer to “is NH₃ a strong base?” provides a gateway to deeper scientific insight.
Conclusion
So, is NH₃ a strong base? The definitive answer is no — ammonia is a weak base in aqueous solution, with a Kb of about 1.8 × 10⁻⁵ and a pKb of roughly 4.75. Its basicity is modest compared to the near‑complete dissociation of NaOH or KOH, yet it remains profoundly important across industries, biology, and everyday life. The distinction hinges on how completely ammonia ionizes, the equilibrium constants involved, and the environmental factors that can shift its behavior. Armed with this knowledge, you can make informed decisions — from safe handling in the home to strategic use in industrial applications. Embrace the subtlety of ammonia’s chemistry, and you’ll find that even a “weak” base can wield a surprisingly strong impact.