Boron Lewis Dot Structure: Why Boron Defies The Octet Rule
Have you ever wondered why some elements, like boron, seem to break the fundamental rules of chemical bonding we learn in introductory chemistry? The boron Lewis dot structure is a fascinating exception that challenges the iconic octet rule, revealing the nuanced and elegant complexity of atomic interactions. While most main group elements strive for a stable configuration of eight valence electrons, boron proudly stands apart, often content with just six. This unique behavior isn't a mistake; it's a defining characteristic that makes boron indispensable in everything from specialized glasses to cutting-edge pharmaceuticals. Understanding this deviation is crucial for anyone looking to grasp the deeper principles of chemical reactivity and molecular design. So, let's pull back the curtain on boron's electron-deficient world and explore what its simple dot structure truly signifies.
The Atomic Blueprint: Boron's Electron Configuration
To understand the boron Lewis dot structure, we must first return to its atomic foundation. Boron, with the atomic number 5, has an electron configuration of 1s²2s²2p¹. This means its two innermost electrons are tightly held in the first shell (the K-shell), completely filling it. The chemical action happens in the second, valence shell (the L-shell), which contains the remaining three electrons: two paired in the 2s orbital and one unpaired in a 2p orbital.
This distribution—three valence electrons—is the starting point for constructing its Lewis symbol. In Lewis notation, we represent an element's symbol surrounded by dots, each representing one valence electron. For boron, this is simply B with three dots placed around it, typically one on each of three sides to maximize separation according to Hund's rule. This minimalist arrangement immediately hints at boron's limitation: with only three electrons to share, it can form, at most, three covalent bonds by sharing each of its valence electrons with another atom.
The Sacred Octet Rule and Its Most Famous Defector
The octet rule is the cornerstone of introductory bonding theory. It states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons, mirroring the stable electron configuration of noble gases. For elements like carbon, nitrogen, oxygen, and fluorine, this rule is a reliable predictor of molecular geometry and stability. Boron, sitting in Group 13 (or IIIA), is the first element in the p-block where this rule begins to fray.
With its three valence electrons, if boron were to form three single covalent bonds (as it commonly does), it would only share six electrons in total. In a molecule like boron trifluoride (BF₃), boron is the central atom bonded to three fluorine atoms. Each B-F bond is a single covalent bond where boron contributes one electron and fluorine contributes one. After forming these three bonds, boron has six electrons in its valence shell—two from each bond it shares. It has not achieved an octet; it has an incomplete octet or is electron-deficient. This is not a temporary state; for many boron compounds, six is a stable, ground-state configuration. Boron is small, has a relatively high ionization energy, and lacks a low-lying d-orbital set to easily expand its octet like elements in Period 3 and beyond. Its path to stability is different.
Boron Compounds with Six Valence Electrons: Embracing Deficiency
The most common and stable boron compounds feature boron with a formal oxidation state of +3 and exactly six valence electrons. This isn't a rare anomaly; it's the norm. Boron trihalides (BF₃, BCl₃, BBr₃, BI₃) and borane (BH₃, though it typically dimerizes to B₂H₆) are prime examples. Why is a configuration with only six electrons stable for boron?
The answer lies in a combination of factors. First, the energetic cost of promoting an electron from the 2s to a 2p orbital to form four sp³ hybrid orbitals (which would allow four bonds and an octet) is high for a small atom like boron. The energy gained by forming a fourth bond often doesn't compensate for this promotion cost. Second, the six-electron, trigonal planar geometry (sp² hybridization) is geometrically efficient and allows for optimal orbital overlap with its bonding partners. Third, and most importantly, this electron deficiency makes boron compounds powerful Lewis acids, giving them a unique and highly useful chemical personality. They are not "incomplete" in a pathological sense; they are designed by nature to be electron acceptors.
Beyond Trihalides: A World of Electron-Deficient Species
While BF₃ is the classic example, the theme of six valence electrons extends widely. The molecule BH₃ itself is a monomer with an incomplete octet, but it is so reactive that it instantly dimerizes to form diborane (B₂H₆), which features fascinating multi-center bonds. In boric acid (B(OH)₃), boron is bonded to three oxygen atoms and also has only six valence electrons. Here, the oxygen atoms are bonded to hydrogen, but the boron center remains electron-deficient. This deficiency is why boric acid acts as a weak Lewis acid in water, accepting a hydroxide ion to form the tetrahydroxyborate ion, [B(OH)₄]⁻, where boron finally achieves an octet. This pattern—a stable six-electron species that can accept an electron pair to become an eight-electron species—is a recurring theme in boron chemistry.
Boron Trifluoride: The Poster Child for Electron Deficiency
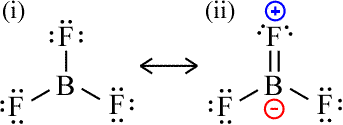
No discussion of the boron Lewis dot structure is complete without a deep dive into boron trifluoride (BF₃). It is the archetypal electron-deficient molecule and a cornerstone example in chemistry textbooks. Its Lewis structure is deceptively simple: a boron atom in the center with single bonds to three fluorine atoms, and no lone pairs on boron. This gives boron a formal count of six valence electrons.
The molecular geometry is trigonal planar, with bond angles of 120°. This geometry arises from sp² hybridization of the boron atomic orbitals, where three sp² hybrid orbitals form sigma bonds with the fluorine atoms, and the remaining unhybridized p orbital (perpendicular to the plane) is empty. This empty p orbital is the key to BF₃'s reactivity. It creates a strong electrostatic partial positive charge on the boron atom, making it a powerful Lewis acid. The fluorine atoms, being highly electronegative, pull electron density away from boron, exacerbating this effect.
The Paradox of BF₃'s Stability
Given its electron deficiency, why doesn't BF₃ immediately react with everything to complete its octet? The stability of BF₃ is a balance. The B-F bonds are exceptionally strong due to the excellent overlap between the small boron 2p orbital and the fluorine 2p orbital, and significant back-bonding from fluorine's lone pairs into boron's empty p orbital (a phenomenon called pπ-pπ back-bonding). This back-bonding provides some electron density to boron, mitigating its deficiency and stabilizing the molecule. However, this stabilization is not enough to quench its Lewis acidity; it merely modulates it. BF₃ is a gas at room temperature and is commercially available, but it is notoriously reactive, fuming in moist air and vigorously attacking many materials. Its Lewis structure perfectly predicts this dual nature: stable enough to isolate, yet hungry for electrons.
Boron as a Lewis Acid: The Quest for Completeness
The defining chemical behavior stemming from the boron Lewis dot structure is its role as a Lewis acid—an electron-pair acceptor. This is the direct consequence of having an incomplete octet and an accessible, empty orbital. When a Lewis base (an electron-pair donor, like ammonia, NH₃, or fluoride ion, F⁻) approaches boron, it can donate its lone pair into boron's empty orbital, forming a new coordinate covalent bond (also called a dative bond).
This process transforms boron's electron count. In the adduct BF₃·NH₃ (fluoroborane-ammonia adduct), boron now has four bonds: three to fluorine and one to nitrogen via the donated pair. Its Lewis structure now shows four bonding pairs around boron—eight valence electrons. It has achieved an octet. The geometry changes from trigonal planar to tetrahedral around boron, reflecting sp³ hybridization. This ability to seamlessly transition from a six-electron to an eight-electron species is central to boron's utility in synthesis.
Common Lewis Acid-Base Reactions Involving Boron
This Lewis acid behavior is ubiquitous:
- With Halide Ions: BF₃ + F⁻ → BF₄⁻ (tetrafluoroborate ion). This reaction is so favorable that BF₃ is often used as a fluoride ion scavenger in organic synthesis.
- With Ethers: BF₃ forms stable 1:1 adducts with diethyl ether (BF₃·OEt₂), a common laboratory complex.
- With Water: BF₃ hydrolyzes: BF₃ + 3H₂O → B(OH)₃ + 3HF. Here, water acts as a base, donating electron pairs to boron, ultimately leading to boric acid.
- In Organic Synthesis: Boron trifluoride etherate (BF₃·OEt₂) is a vital catalyst for Friedel-Crafts acylation and alkylation, polymerization, and isomerization reactions. It activates substrates by coordinating to electron-rich atoms like oxygen.
Practical Implications: Why Boron's Quirk Matters
Understanding the boron Lewis dot structure is not an academic exercise. Its electron-deficient nature is the source of its immense practical value across multiple scientific fields.
In Organic Synthesis: Precision Tools
Boron reagents are workhorses in modern organic chemistry. The hydroboration reaction, using borane (BH₃) or its derivatives, allows for the anti-Markovnikov, syn addition of hydrogen and boron across alkenes. This reaction is stereospecific and provides a route to alcohols after oxidation. The driving force is boron's desire to accept electron density from the alkene's π-bond. Similarly, diborane (B₂H₆), with its unique banana bonds, is a precursor to countless boron-containing compounds. The Suzuki-Miyaura cross-coupling reaction, which won the 2010 Nobel Prize in Chemistry, relies on organoboron compounds (like arylboronic acids) as coupling partners. The boron-carbon bond is stable yet reactive under palladium catalysis, a direct consequence of boron's electronic character.
In Materials Science: Strength and Novelty
Boron's ability to form strong, covalent networks is exploited in borosilicate glass (e.g., Pyrex), where boron oxide (B₂O₃) introduces thermal shock resistance. The boron atoms in this glass network are typically in a triangular coordination with oxygen, consistent with its sp² hybridization and six-electron environment. Boron nitride (BN) exists in two primary forms: a graphite-like hexagonal form (h-BN), a excellent lubricant and insulator, and a diamond-like cubic form (c-BN), second only to diamond in hardness. In h-BN, boron and nitrogen alternate in a planar sheet, with boron's electron deficiency contributing to the layer structure. Boron carbide (B₄C) is one of the hardest known materials, used in armor and abrasive applications, its extreme hardness stemming from a complex, strong covalent network built around boron icosahedra.
In Biology and Medicine: A Trace Element with Major Impact
Boron is a essential trace element for plants, influencing cell wall formation, sugar transport, and hormone regulation. Its mechanism likely involves forming complexes with biomolecules like sugars, leveraging its Lewis acidity. In human health, boron's role is less defined but potentially significant for bone density and cognitive function. Boron-containing compounds are also in medical use; boronic acid derivatives are a key class of drugs, most notably in the proteasome inhibitor bortezomib (Velcade®), used to treat multiple myeloma. The boronic acid group reversibly binds to the active site of the proteasome enzyme, a process facilitated by boron's ability to form tetrahedral, octet-complete intermediates.
Addressing Common Questions: Boron Lewis Structure FAQs
Q: Can boron ever have more than eight electrons?
A: Yes, but it's rare and requires special conditions. Boron can expand its octet by utilizing its empty 2d orbitals, but this is energetically unfavorable and not common in simple compounds. Examples are found in some complex fluorides like [BF₄]⁻ (where boron has 8) or in highly coordinated species like [B(OH)₄]⁻. The stable, simple molecules almost always feature boron with 6 or 8 electrons.
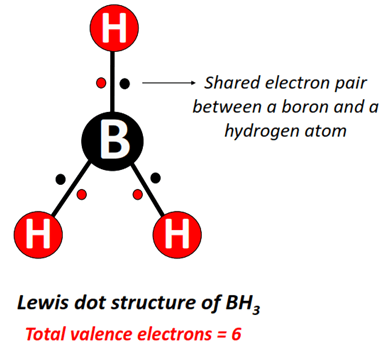
Q: Why doesn't boron form a stable BH₃ molecule?
A: BH₃ is a monomer with an incomplete octet and is extremely reactive. To reduce its electron deficiency, two BH₃ units combine to form diborane (B₂H₆). In B₂H₆, boron achieves a sort of "electron sharing" through two three-center two-electron bonds (banana bonds), where a pair of electrons is shared between three atoms (B-H-B). This is a different bonding paradigm entirely, born from boron's electron deficiency.
Q: Is the boron Lewis dot structure always with three bonds?
A: For neutral boron compounds in its +3 oxidation state, yes, three bonds and six electrons is the standard. However, in anionic species like BF₄⁻ or B(OH)₄⁻, boron forms four bonds and has a complete octet. The Lewis structure must reflect the formal charge and total valence electron count.
Q: How do I draw the Lewis structure for a boron compound?
Actionable Tip:
- Count Total Valence Electrons: Sum the valence electrons of all atoms. For BF₃: B (3) + 3*F (7 each) = 24 electrons.
- Place Boron as Central Atom: Boron is less electronegative than fluorine.
- Connect with Single Bonds: Use 6 electrons to form three B-F bonds.
- Complete Octets on Terminal Atoms: Distribute the remaining 18 electrons to the fluorines as lone pairs (each F gets 6 more electrons, 3 lone pairs). This uses all 24 electrons.
- Check the Central Atom: Boron now has 6 electrons (three bonds). This is acceptable. Do not add more electrons or bonds to force an octet on boron. The structure is correct with an incomplete octet.
Conclusion: Embracing the Exception
The boron Lewis dot structure is a masterclass in chemical exception that proves the rule. Its simple depiction—a boron atom with three bonds and no lone pairs—belies a profound truth: stability in chemistry is not a one-size-fits-all concept. Boron's electron-deficient nature, manifesting as an incomplete octet, is not a flaw but a fundamental feature. It is the engine behind boron's unparalleled versatility as a Lewis acid, its crucial role in synthetic methodologies that build modern medicines, and its contribution to materials with extraordinary thermal and mechanical properties.
From the trigonal planar geometry of BF₃ to the tetrahedral [BF₄]⁻ ion, boron seamlessly navigates between six and eight electrons, accepting and donating electron pairs with precision. This behavior challenges novice chemists to think beyond the octet rule and appreciate the deeper quantum mechanical principles—orbital hybridization, electronegativity, and back-bonding—that govern molecular architecture. The next time you see a simple Lewis structure for a boron compound, remember: you are not looking at an incomplete molecule, but at a chemically sophisticated one, perfectly adapted to be electron-deficient and powerfully reactive. Understanding this is the key to unlocking boron's vast potential in the laboratory and in industry.