CH2Cl2 Polar Or Nonpolar: Understanding Dichloromethane's Molecular Nature
Have you ever wondered why some substances dissolve in water while others don't? Or why certain chemicals mix well while others remain separate? The answer often lies in a fundamental property called polarity. Today, we're diving deep into the question: CH2Cl2 polar or nonpolar? This seemingly simple question opens up a fascinating world of molecular geometry, electronegativity, and chemical behavior.
Dichloromethane, with the chemical formula CH2Cl2, is a common organic solvent used in various industries. But before we can understand its properties and applications, we need to determine whether it's polar or nonpolar. This classification affects everything from its solubility to its reactivity and environmental impact. So, let's unravel the mystery of CH2Cl2's polarity and explore what makes this molecule tick.
The Basics of Molecular Polarity
To determine if CH2Cl2 is polar or nonpolar, we first need to understand what molecular polarity means. Polarity refers to the distribution of electrical charge across a molecule. In a polar molecule, there's an uneven distribution of electrons, creating regions of partial positive and negative charges. Nonpolar molecules, on the other hand, have an even distribution of electrons.
The polarity of a molecule depends on two main factors: the polarity of its individual bonds and its molecular geometry. Even if a molecule contains polar bonds, its overall shape can sometimes cancel out these polarities, resulting in a nonpolar molecule. This is where the concept of molecular geometry becomes crucial.
CH2Cl2 Structure and Composition
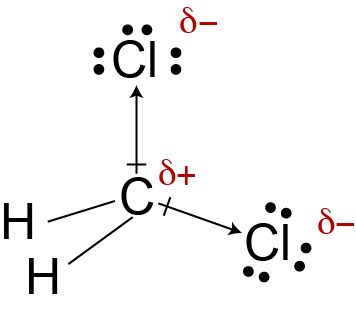
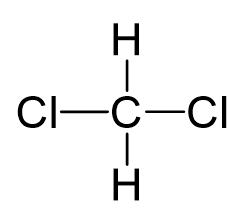
CH2Cl2, also known as dichloromethane, consists of one carbon atom, two hydrogen atoms, and two chlorine atoms. The carbon atom forms four single bonds - two with hydrogen atoms and two with chlorine atoms. This arrangement gives CH2Cl2 a tetrahedral molecular geometry.
The tetrahedral shape is important because it affects how the molecule's polarities interact. In a perfect tetrahedron, if all four surrounding atoms were identical, the polarities would cancel out, resulting in a nonpolar molecule. However, CH2Cl2 doesn't have identical atoms surrounding the central carbon, which is key to understanding its polarity.
Electronegativity and Bond Polarity
To further understand CH2Cl2's polarity, we need to consider electronegativity. This is a measure of an atom's ability to attract electrons in a chemical bond. The difference in electronegativity between bonded atoms determines the polarity of that bond.
In CH2Cl2:
- Carbon has an electronegativity of about 2.5
- Hydrogen has an electronegativity of about 2.1
- Chlorine has an electronegativity of about 3.0
The difference between carbon and hydrogen is relatively small (0.4), making the C-H bonds slightly polar. However, the difference between carbon and chlorine is more significant (0.5), making the C-Cl bonds more polar.
Is CH2Cl2 Polar or Nonpolar? The Verdict
After considering all factors, we can confidently say that CH2Cl2 is polar. Despite its tetrahedral geometry, the molecule's asymmetry - with two different types of atoms attached to the central carbon - prevents the bond dipoles from canceling out completely.
The two C-Cl bonds, being more polar than the C-H bonds, create a net dipole moment. This means that CH2Cl2 has a slight positive charge on the side with the hydrogen atoms and a slight negative charge on the side with the chlorine atoms. This asymmetric distribution of charge is what makes CH2Cl2 a polar molecule.
Implications of CH2Cl2's Polarity
The polarity of CH2Cl2 has significant implications for its properties and uses:
Solubility: As a polar molecule, CH2Cl2 can dissolve many polar and ionic compounds. This makes it an excellent solvent in various applications, from paint stripping to pharmaceutical manufacturing.
Boiling Point: Polar molecules generally have higher boiling points than nonpolar molecules of similar size due to stronger intermolecular forces. CH2Cl2 has a boiling point of about 40°C, which is relatively high for its molecular weight.
Reactivity: The polarity of CH2Cl2 affects its chemical reactivity. Polar molecules are often more reactive than nonpolar ones because the uneven charge distribution can make certain parts of the molecule more susceptible to chemical attacks.
Environmental Impact: The polarity of CH2Cl2 influences how it behaves in the environment. Polar compounds are generally more soluble in water and can be more mobile in soil and groundwater.
Comparing CH2Cl2 to Other Molecules
To better understand CH2Cl2's polarity, it's helpful to compare it to similar molecules:
CH4 (Methane): This molecule is nonpolar because all four surrounding atoms are identical hydrogen atoms, resulting in a symmetrical charge distribution.
CH3Cl (Chloromethane): Like CH2Cl2, this molecule is polar due to the presence of different atoms around the central carbon.
CCl4 (Carbon Tetrachloride): Despite having polar C-Cl bonds, this molecule is nonpolar because its symmetrical tetrahedral shape allows the bond dipoles to cancel out.
These comparisons highlight how molecular geometry and the types of atoms involved can significantly impact a molecule's overall polarity.
Practical Applications of CH2Cl2's Polarity
The polar nature of CH2Cl2 makes it valuable in numerous applications:
Solvent in Chemical Reactions: Its ability to dissolve both polar and nonpolar substances makes it a versatile solvent in organic chemistry.
Paint Remover: CH2Cl2's polarity allows it to break down and dissolve various types of paint and varnish.
Pharmaceutical Industry: It's used in the production of various drugs due to its ability to extract and purify compounds.
Food Industry: In small quantities, it's used to decaffeinate coffee and tea.
Metal Cleaning: Its polarity helps in removing oils and greases from metal surfaces.
Safety Considerations for CH2Cl2
While CH2Cl2's polarity makes it useful, it's important to note that it can be hazardous:
- It's volatile and can form explosive mixtures with air.
- Inhalation can cause dizziness, nausea, and in severe cases, unconsciousness or death.
- It's a potential carcinogen and can harm the central nervous system.
- Proper ventilation and personal protective equipment are essential when handling CH2Cl2.
Conclusion
In conclusion, CH2Cl2 is indeed a polar molecule. Its polarity arises from the combination of polar C-Cl and C-H bonds and its asymmetrical tetrahedral structure. This polarity gives CH2Cl2 its unique properties and makes it valuable in various industrial and laboratory applications. However, it also necessitates careful handling due to its potential health and environmental impacts.
Understanding the polarity of molecules like CH2Cl2 is crucial in chemistry, as it influences everything from a substance's physical properties to its behavior in chemical reactions and the environment. As we continue to explore the world of chemistry, questions like "CH2Cl2 polar or nonpolar?" remind us of the intricate relationships between molecular structure and chemical properties.