Does Facilitated Diffusion Require Energy? The Surprising Truth About Passive Cellular Transport
Does facilitated diffusion require energy? It’s a deceptively simple question that sits at the heart of understanding how our cells function. For anyone who has ever studied biology, the maze of cellular transport mechanisms can be confusing. You hear terms like "active transport" and "passive transport," and it’s easy to assume that any process involving a protein "pump" must be burning cellular fuel. But here’s the counterintuitive truth: facilitated diffusion is a passive process. It does not require the cell to expend ATP or any other direct form of metabolic energy. Instead, it cleverly harnesses the natural, relentless power of a concentration gradient to move vital substances where they’re needed. This isn't just a minor textbook detail; it’s a fundamental principle that explains everything from how your nerves fire to how your kidneys filter blood. In this deep dive, we’ll dismantle the myths, explore the molecular machinery, and definitively answer whether facilitated diffusion requires energy.
What Is Facilitated Diffusion? A Passive Transport Primer
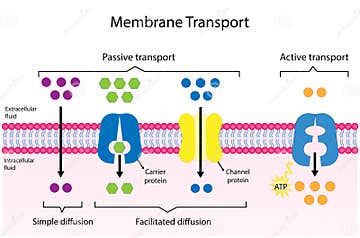
At its core, facilitated diffusion is a type of passive transport. This means the movement of molecules across a cell membrane occurs spontaneously, driven solely by the difference in concentration on either side of that membrane. Think of it like opening a window on a windy day—air rushes from the high-pressure zone (inside) to the low-pressure zone (outside) without you having to blow. Similarly, molecules in facilitated diffusion move down their concentration gradient, from an area of higher concentration to an area of lower concentration. The "facilitated" part comes from the fact that certain molecules—typically large, polar, or charged ones like glucose, amino acids, and ions—cannot simply dissolve through the hydrophobic (water-fearing) lipid bilayer on their own. They need help.
This help comes in the form of specific integral membrane proteins embedded in the cell membrane. These proteins act as selective gateways or shuttles. They are not generic doors; they are highly specific, often recognizing and transporting only one type of molecule or a very small group of similar molecules. This specificity is crucial for cellular order. Without it, a neuron couldn't maintain its electrochemical gradient, and a liver cell couldn't absorb glucose efficiently from the bloodstream. The process is also saturable, meaning there’s a maximum rate at which it can occur. Once all the protein transporters are occupied, increasing the concentration gradient won’t speed things up further—a key feature distinguishing it from simple diffusion.
Why Cells Need Facilitated Diffusion: The Problem with the Lipid Bilayer
To understand why this process exists, we must first appreciate the barrier it overcomes: the phospholipid bilayer. This bilayer is the foundational structure of all cell membranes, a clever arrangement where hydrophilic (water-loving) heads face outward and hydrophobic (water-fearing) tails face inward, creating a oily, impermeable core. Small, nonpolar molecules like oxygen (O₂) and carbon dioxide (CO₂) can slip through this core with ease via simple diffusion. So can small, uncharged molecules like water (though water often uses specialized channels called aquaporins for speed). However, the molecules a cell desperately needs to import—like glucose for energy or sodium ions for signaling—are often polar or charged. They are repelled by the hydrophobic interior, much like a magnet repels another magnet with the same pole.
This is where facilitated diffusion becomes a biological necessity. Imagine trying to get a crowd of people (polar molecules) across a moat (the lipid bilayer) without a bridge. They’re stuck. The cell, therefore, evolved to build specialized bridges: transport proteins. These proteins provide a hydrophilic pathway through the membrane, a tunnel or a binding site that shields the polar molecule from the oily interior. Without this mechanism, essential nutrients and signaling ions would be locked outside the cell, and metabolic waste products would build up inside. Life, as we know it, would simply not be possible. It’s a perfect example of evolution solving a physical-chemical problem with a molecular machine.
The Energy Question: Why Facilitated Diffusion Is Truly Passive
So, does facilitated diffusion require energy? The definitive, evidence-based answer is no. This is the most critical point to grasp. The confusion usually stems from observing that carrier proteins change shape during transport. This conformational change can look active, like a machine moving parts. However, the energy for this shape shift does not come from ATP hydrolysis. Instead, it comes directly from the binding energy of the molecule itself and the thermodynamic drive of the concentration gradient.
Here’s the step-by-step for a carrier protein like the GLUT1 glucose transporter:
- The transporter has a binding site for glucose on the outside of the membrane (side with higher glucose concentration).
- Glucose binds to this site. This binding event has a specific affinity and releases a tiny amount of energy.
- This binding energy triggers a conformational change—the protein reshapes—now exposing the bound glucose to the inside of the cell (side with lower glucose concentration).
- Glucose dissociates into the cytoplasm, and the transporter returns to its original shape, ready for another cycle.
At no point is ATP or any other nucleoside triphosphate (like GTP) hydrolyzed to power this cycle. The entire process is driven by the spontaneous tendency of glucose to move from high to low concentration. The protein is a catalyst and a guide, not an engine. Channel proteins, which form open pores, are even more straightforward. They are either always open (leak channels) or gated open by voltage or ligand binding, but the ion flow itself is a passive diffusion event through the aqueous pore, governed entirely by electrochemical gradients. The cell conserves immense amounts of energy by using passive transport for substances where a gradient already exists.
Carrier Proteins vs. Channel Proteins: Two Mechanisms, One Goal
Facilitated diffusion employs two distinct, yet complementary, classes of transport proteins: carrier proteins (also called transporters) and channel proteins. Understanding their differences clarifies why no external energy is needed.
Carrier Proteins work via a "alternating access" model, as described above. They bind their specific substrate on one side, undergo a dramatic conformational change, and release it on the other side. This mechanism is highly specific and can be regulated. For example, insulin signaling increases the number of GLUT4 transporters on muscle and fat cell membranes, enhancing glucose uptake without any change in ATP use for the transport itself—the regulation happens at the level of protein trafficking. Carrier proteins are saturable and can be inhibited by competitive molecules that bind to their site.
Channel Proteins, in contrast, form a hydrophilic pore that is continuously open or gated. They do not bind their substrate in a one-to-one manner; instead, ions or water molecules flow through the pore in single file, driven by their electrochemical gradient. Think of it like a crowded hallway—people (ions) can move through quickly if the hallway is open. Ion channels (e.g., potassium leak channels, voltage-gated sodium channels) are crucial for generating electrical signals in neurons. Aquaporins are specialized water channels that allow rapid water movement, essential for kidney function and plant water uptake. Channel opening can be triggered by membrane voltage (voltage-gated), ligand binding (ligand-gated), or mechanical stretch (mechanosensitive). The flow through an open channel is entirely passive and can be incredibly rapid, moving millions of ions per second.
Real-Life Examples: From Glucose to Nerve Signals
The abstract concept of facilitated diffusion springs to life when we look at its concrete roles in human physiology.
- Glucose Uptake: This is the classic example. After you eat, blood glucose rises. Cells in your muscles, fat, and brain express GLUT transporters (GLUT1, GLUT3, GLUT4) to bring glucose inside for energy production. GLUT4 is insulin-sensitive, which is why this process is defective in type 2 diabetes. The glucose moves into the cell down its concentration gradient, facilitated by the carrier protein—no ATP spent by the transporter.
- Neuronal Signaling: Your brain's electrical chatter relies on facilitated diffusion. At rest, neurons maintain a high internal concentration of potassium ions (K⁺) and a low internal concentration of sodium ions (Na⁺). Leak channels for K⁺ allow potassium to passively diffuse out of the cell down its electrochemical gradient, creating a negative resting potential. During an action potential, voltage-gated sodium channels open, allowing Na⁺ to rush in passively, depolarizing the membrane. This influx is the initial spark of the nerve signal.
- Kidney Function: In the nephrons of your kidneys, facilitated diffusion reabsorbs vital ions. For instance, in the distal convoluted tubule, a sodium-chloride cotransporter (a carrier) moves both ions from the urine back into the blood, down their combined concentration gradient. This is a passive step in a larger active process.
- Water Balance:Aquaporin-2 channels in kidney collecting duct cells are inserted in response to the hormone vasopressin (ADH). When open, they allow massive amounts of water to follow the osmotic gradient created by active sodium reabsorption, concentrating your urine. The water movement itself is passive facilitated diffusion.
These examples show that this process isn't a minor sidebar; it's central to metabolism, communication, and homeostasis.
Facilitated Diffusion vs. Active Transport: A Clear Comparison
The confusion between facilitated diffusion and active transport is the root of the "does it require energy?" question. Let’s lay them side-by-side to eliminate doubt.
| Feature | Facilitated Diffusion | Active Transport |
|---|---|---|
| Energy Requirement | No. No direct ATP or metabolic energy used. | Yes. Requires ATP hydrolysis or an electrochemical gradient (secondary active). |
| Direction of Movement | Down the concentration/electrochemical gradient. | Against the concentration/electrochemical gradient. |
| Key Proteins | Channel proteins & Carrier proteins (transporters). | Pumps (e.g., Na⁺/K⁺-ATPase, H⁺-ATPase). |
| Saturation | Yes. Limited by number of transport proteins. | Yes. Limited by number of pump proteins. |
| Specificity | High. Usually specific for one ion or molecule type. | High. Pumps are highly specific. |
| Example | Glucose entry via GLUT4; K⁺ leak from a neuron. | Sodium-potassium pump; Proton pump in stomach lining. |
The takeaway is stark: if a substance is moving up its gradient, the cell is paying an energy cost (active transport). If it’s moving down its gradient, even with a protein’s help, it’s a passive, energy-free process (facilitated diffusion). The Na⁺/K⁺-ATPase pump is the quintessential active transporter—it uses one ATP to move 3 Na⁺ out and 2 K⁺ in, against their gradients. The potassium leak channel that lets K⁺ diffuse out is facilitated diffusion.
What Affects the Rate of Facilitated Diffusion?
While facilitated diffusion doesn’t require energy, its rate is not infinite. It’s influenced by several physical and biological factors:
- Steepness of the Concentration Gradient: A larger difference in concentration across the membrane results in a faster net movement, up to the point where all transporters are saturated.
- Number of Transport Proteins: More channels or carriers in the membrane mean more "highways" for the substance to use. Cells can regulate this by inserting or removing proteins from the membrane (e.g., GLUT4 translocation in muscle cells).
- Saturation Kinetics: Unlike simple diffusion, which increases linearly with concentration, facilitated diffusion follows a curve described by the Michaelis-Menten equation (borrowed from enzyme kinetics). At low substrate concentrations, rate increases sharply with concentration. At high concentrations, all transporters are occupied (Vmax is reached), and the rate plateaus.
- Temperature: Increased temperature generally increases the rate of molecular motion and the fluidity of the membrane, speeding up the process up to a point where proteins denature.
- Ion Concentration & Membrane Potential: For charged particles (ions), the electrochemical gradient is key. This combines the chemical concentration gradient with the electrical gradient (membrane potential). For example, sodium ions (Na⁺) are both concentrated outside and attracted to the negative interior, creating a powerful driving force for their passive influx through channels.
Understanding these factors helps explain cellular responses. When you exercise and adrenaline surges, your muscle cells rapidly increase glucose uptake not by making more GLUT4 from scratch, but by trafficking existing GLUT4 vesicles to the membrane, instantly increasing the number of transporters and thus the facilitated diffusion rate.
Debunking Common Myths About Facilitated Diffusion
Myths about this process persist, especially the one linking conformational change to energy use.
Myth 1: "The carrier protein changes shape, so it must use ATP."
Reality: The conformational change in a carrier protein is entropically and enthalpically driven by the binding of the substrate molecule and the thermodynamic imperative to equalize concentrations. The energy comes from the spontaneous binding reaction and the gradient itself, not from ATP hydrolysis. It’s a passive, downhill process.
Myth 2: "Facilitated diffusion is just a fancy name for active transport."
Reality: They are fundamentally opposite. Active transport moves against the gradient and requires energy. Facilitated diffusion moves with the gradient and does not. Confusing them leads to a catastrophic misunderstanding of cellular energetics.
Myth 3: "All transport through proteins is active."
Reality: This is a classic overgeneralization. While pumps are active, channels and many transporters (like GLUTs) are passive. The protein’s function, not its mere existence, determines the energy requirement.
Myth 4: "Facilitated diffusion is slow."
Reality: It can be extremely fast. Ion channels can move 10⁷ to 10⁸ ions per second. While slower than channels, carrier-mediated transport is still orders of magnitude faster than simple diffusion for large polar molecules. Speed is relative to the alternative (no transport).
Myth 5: "Cells use facilitated diffusion for everything."
Reality: Cells use it precisely for substances where a gradient exists and passive movement is sufficient. For substances that need to be accumulated against a gradient (like potassium inside a neuron), active transport is mandatory. The two systems work in tandem.
Frequently Asked Questions
Q: Is facilitated diffusion considered active or passive?
A: It is unequivocally a form of passive transport. No cellular energy (ATP) is directly consumed by the transport protein.
Q: Why do cells need facilitated diffusion if simple diffusion exists?
A: Simple diffusion is too slow or impossible for large, polar, or charged molecules (like glucose, ions) because they are repelled by the hydrophobic lipid bilayer. Facilitated diffusion provides a selective, high-capacity pathway for these essential molecules.
Q: Can facilitated diffusion ever move molecules against their concentration gradient?
A: No. By definition, it moves molecules down their electrochemical gradient. Moving against the gradient requires active transport, which uses energy.
Q: What is the difference between a carrier and a channel in facilitated diffusion?
A: Carriers bind specifically to one molecule, change shape, and transport it one at a time (like a ferry). They are saturable and slower. Channels form open pores that allow many ions or molecules to flow through rapidly (like a tunnel), often gated by voltage or ligands.
Q: Is the sodium-potassium pump an example of facilitated diffusion?
A: No. The Na⁺/K⁺-ATPase is the poster child for primary active transport. It uses ATP to pump sodium out and potassium in against their gradients.
Q: Does temperature affect facilitated diffusion?
A: Yes. Increased temperature increases molecular motion and membrane fluidity, generally increasing the rate, up to the point where the transport proteins denature and lose function.
Conclusion: The Elegant Efficiency of Going with the Flow
So, does facilitated diffusion require energy? The resounding answer, backed by decades of molecular biology research, is no. This process is a masterpiece of biological efficiency. Instead of wasting precious ATP to move every molecule, cells exploit the natural law of diffusion—things move from high to low concentration—and simply build the right doors (transport proteins) to let the right substances through. It’s a passive, gradient-driven system that is fundamental to life.
From the glucose fueling your muscles to the potassium ions generating your thoughts, facilitated diffusion operates silently and effortlessly in every cell of your body. Recognizing its passive nature is key to understanding cellular physiology, the action of drugs (many target specific channels or transporters), and diseases like cystic fibrosis (where a defective chloride channel disrupts passive ion flow). The next time you wonder how a cell works, remember: sometimes the smartest strategy isn't to push harder, but to build a better bridge and let nature do the work. That’s the elegant, energy-free truth of facilitated diffusion.