Does Facilitated Diffusion Require Energy? The Surprising Truth About Passive Transport
Does facilitated diffusion require energy? It’s a deceptively simple question that unlocks a fundamental secret of how our cells function. For many of us, the word "diffusion" might conjure an image of a drop of ink spreading in water—a completely passive, energy-free process. But when you add the word "facilitated," does that change the rules? The answer is a resounding no, but the "how" and "why" are where the true cellular magic happens. This isn't just a trivia question for biology students; it's a cornerstone concept that explains everything from how your nerves fire to how your kidneys filter blood. Understanding this process reveals the breathtaking efficiency of life at the microscopic level, where movement happens not through brute force, but through cleverly designed pathways that work with nature, not against it. Let's dive deep into the elegant world of passive transport and settle this question once and for all.
What Exactly Is Facilitated Diffusion? Defining the Process
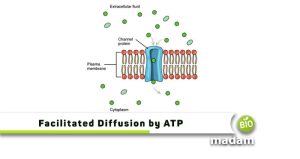
Before we can answer whether it requires energy, we must have an absolutely clear picture of what facilitated diffusion is. At its core, facilitated diffusion is a type of passive transport. This means it moves substances across a cell membrane down their concentration gradient—from an area of higher concentration to an area of lower concentration—without any direct input of cellular energy (like ATP). The "facilitated" part is the crucial twist. It’s used for specific molecules, primarily polar molecules (like glucose) and ions (like sodium, potassium, calcium), that are too large or too charged to slip directly through the hydrophobic (water-repelling) fatty interior of the phospholipid bilayer on their own.
Think of the cell membrane as a guarded, oily wall. Simple, nonpolar molecules like oxygen or carbon dioxide can diffuse right through. But for essential, water-soluble nutrients and signaling ions, that wall is an impassable barrier. This is where transport proteins come in. These are specialized protein molecules embedded in the membrane that act as gatekeepers or tunnels. They provide a hydrophilic (water-loving) pathway, shielding the polar substance from the oily membrane core, allowing it to move across. This process is entirely driven by the inherent kinetic energy of the molecules themselves and the force of the concentration gradient. No cellular "pushing" is involved.
The Two Main Cast Members: Carrier vs. Channel Proteins
There are two primary types of transport proteins involved in facilitated diffusion, and understanding their mechanics is key to seeing why no energy is needed.
1. Carrier Proteins (Transporters): These proteins work through a conformational change. They have a specific binding site for their target molecule (e.g., a glucose transporter for glucose). When the molecule on the high-concentration side binds, the carrier protein changes its shape, enclosing the molecule and then opening to the other side of the membrane, releasing it. It’s like a revolving door that only turns when someone (the molecule) is on one side, and it turns because of the pressure from the crowd (the concentration gradient), not because someone is manually pushing it. The GLUT (Glucose Transporter) family is a classic example, moving glucose into cells after a meal when blood glucose is high.
2. Channel Proteins: These form hydrophilic pores or tunnels that are always open or, more commonly, gated (they open and close in response to a signal, like a voltage change or a chemical ligand). Ions like Na⁺, K⁺, Cl⁻, and Ca²⁺ diffuse through these channels. The driving force is purely the electrochemical gradient—a combination of the concentration difference and the electrical charge difference across the membrane. For instance, potassium leak channels allow K⁺ ions to passively diffuse out of a resting neuron, helping establish the negative internal charge. The opening is triggered, but the movement through the open pore is entirely gradient-driven.
The Concentration Gradient: The True Engine of Movement
This is the most critical concept to grasp. A concentration gradient is a difference in the concentration of a substance across a space—in this case, across the membrane. Molecules are in constant, random motion (Brownian motion). Where there are more molecules (high concentration), there are more collisions and a higher statistical probability that some will move toward the area with fewer molecules (low concentration). This random movement, when summed across trillions of molecules, results in a net flow down the gradient.
Facilitated diffusion does not create this gradient; it simply provides a shortcut for molecules to follow it. The cell expends energy (often via the sodium-potassium pump, an active transport mechanism) to establish certain gradients, like a high concentration of sodium outside and potassium inside. Once that gradient is set up, facilitated diffusion channels for sodium or potassium can allow ions to move passively back down that pre-established hill. The energy was used upfront to build the gradient, not for the facilitated diffusion event itself. It’s like using energy to pump water uphill to create a reservoir; the subsequent flow downhill through a channel (facilitated diffusion) releases that stored potential energy without additional work.
Contrasting with Active Transport: Where the Energy Is Spent
To definitively answer "does facilitated diffusion require energy?", we must contrast it sharply with its energetic cousin, active transport. Active transport moves substances against their concentration gradient—from low to high concentration. This is like pushing water uphill, which absolutely requires an input of energy, almost always from the hydrolysis of ATP (adenosine triphosphate).
- The Sodium-Potassium Pump (Na⁺/K⁺-ATPase): This is the poster child for active transport. For every ATP molecule used, it pumps 3 sodium ions out of the cell and 2 potassium ions into the cell, both against their gradients. This maintains the crucial electrochemical gradient for nerve impulses and cell volume.
- Proton Pumps (H⁺-ATPases): Found in stomach lining cells to create acidic gastric juice, and in plant cell membranes, they use ATP to pump protons out, creating a proton gradient used for other processes.
- Co-transport (Symport/Antiport): Sometimes, the energy from one substance moving down its gradient (often sodium, established by the Na⁺/K⁺ pump) is used to pull another substance against its gradient. While this couples to a gradient created by active transport, the co-transport step itself is often considered a form of secondary active transport because the net movement of the target molecule is against its own gradient.
The key differentiator: If the net movement of the target molecule is down its own electrochemical gradient, and the transport protein is simply providing a pathway, it is facilitated diffusion (passive). If the net movement is up its gradient, it is active transport (energy-requiring), even if it uses a gradient set up by another pump.
Practical Examples: Facilitated Diffusion All Around You
Let’s make this concrete. Facilitated diffusion isn't an abstract concept; it's happening constantly in your body.
- Glucose Uptake in Muscles and Fat Cells: After you eat, blood glucose rises. Muscle and adipose (fat) cells express GLUT4 transporters. Insulin signals these cells to bring more GLUT4 transporters to the membrane. Once there, glucose flows into the cell down its concentration gradient via facilitated diffusion, providing fuel. No ATP is spent for the glucose crossing; the energy cost was in making and trafficking the GLUT4 proteins and in the insulin signaling pathway.
- Neuronal Signaling - The Refractory Period: After a nerve fires, potassium ions need to leave the cell to repolarize the membrane. Delayed rectifier potassium channels open, and K⁺ rushes out down its electrochemical gradient via facilitated diffusion. This is a passive process that is absolutely critical for resetting the neuron.
- Kidney Function - Reabsorption: In the kidney tubules, vital substances like glucose and amino acids are reabsorbed from the filtrate back into the blood. This is primarily done via sodium-dependent co-transporters (secondary active transport), but the final step for some ions and water involves passive channels and pores, utilizing gradients established by active pumps.
- Plant Nutrient Uptake: Root hair cells use facilitated diffusion channels to take up water and certain mineral ions from the soil when soil concentrations are higher than inside the cell.
Addressing the Common Misconceptions and FAQs
Q: "But don't carrier proteins change shape? Doesn't that take energy?"
A: This is the most common point of confusion. The conformational change in a carrier protein during facilitated diffusion is passively driven by the binding and release of the substrate molecule and the gradient itself. It’s like a spring-loaded door that opens when pushed from one side and closes when the pressure is released. No ATP hydrolysis is involved in the shape change. In contrast, active transport pumps (like the Na⁺/K⁺ pump) undergo an ATP-driven phosphorylation cycle that forces a shape change against the gradient.
Q: "Is all diffusion passive?"
A: Yes, by definition. "Diffusion" implies movement down a gradient. "Facilitated" just specifies the mechanism (using a protein). "Active transport" is a separate category. The terms are not interchangeable.
Q: "Can facilitated diffusion ever become active?"
A: The mechanism itself is inherently passive. However, the cell can regulate it. By inserting or removing transport proteins from the membrane (like GLUT4 with insulin), the cell controls the rate of passive diffusion. The energy is spent on the regulation (protein synthesis, vesicle trafficking), not on the transport event per se.
Q: What about gated channels? Opening a gate must cost energy, right?"
A: The gating mechanism (the opening/closing) can be triggered by energy-using signals (like a phosphorylation event from an ATP-dependent kinase) or by passive physical changes (voltage-sensing domains). However, once the channel is open, the ion movement through the pore is purely down its electrochemical gradient. The energy cost, if any, is for the gate's trigger, not for the ion's passage.
The Cellular Economy: Why Passive Transport is So Brilliant
The fact that facilitated diffusion is passive is a feature, not a bug. It represents a profound principle of biological efficiency. Cells are masters of energetic economization. They use precious ATP sparingly and strategically. By establishing key electrochemical gradients (using active transport) and then harnessing those gradients to power a vast array of passive movements (via facilitated diffusion and other passive processes), cells create an energy currency system. One ATP-driven pump can set up a gradient that facilitates the passive movement of thousands or millions of ions or molecules. This is the basis of chemiosmosis in mitochondria (where a proton gradient drives ATP synthesis) and nerve impulse propagation. Facilitated diffusion is a workhorse in this elegant system, allowing for rapid, controlled, and energy-free movement of critical substances once the gradient is in place.
Summary: The Definitive Answer and Key Takeaways
So, to return to our central question: Does facilitated diffusion require energy?
No. Facilitated diffusion is a passive process. It does not directly require cellular energy in the form of ATP for the transport of molecules across the membrane.
Here is the essential breakdown:
- It moves substances DOWN their concentration/electrochemical gradient.
- It requires specific transmembrane transport proteins (carriers or channels).
- The driving force is the inherent kinetic energy of the molecules and the gradient itself.
- Any energy expenditure by the cell is indirect: it may be used to establish the gradient (via active transport pumps) or to regulate the number of transport proteins in the membrane, but not for the actual facilitated diffusion event.
Understanding this distinction is fundamental to cellular biology, physiology, and medicine. It explains how nutrients enter cells, how nerves communicate, how kidneys function, and how the incredible efficiency of life at the microscopic scale is achieved. The next time you see a diagram of a cell membrane, remember: not all movement requires a power source. Some of life's most vital processes are beautifully, elegantly passive, working in perfect harmony with the natural laws of physics and chemistry.
{{meta_keyword}}